Boltaron® Antibacterial and Antifungal Properties
Your saved textures:
No Favorites

Boltaron® thermoplastic sheet material does not support the growth of common bacteria and fungus under a variety of conditions. Refer to the results below for specific test results under ASTM G21 and G22 protocol. The tests conclude that Boltaron sheet materials perform well in preventing the growth of bacteria and fungus, even without the addition of an antimicrobial additive.
ASTM G21 is designed for the qualitative determination of mildew (fungus) resistance of synthetic polymeric materials. The method is conducted over a 28 day period by a third party lab, during which Boltaron sheet was placed in petri dishes on nutrient salts agar (in triplicate) and inoculated with the test fungi.
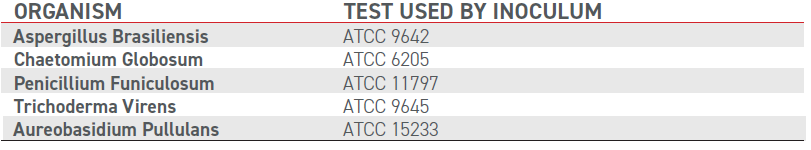
The Boltaron sheet samples did not show any signs of fungus growth after four weeks.
ASTM G22 is a qualitative test method designed to assess the ability of plastics to resist bacterial attack. The method is conducted over a 21 day period during which Boltaron sheet was placed on inoculated agar, incubated, and then compared to a positive and negative control.
The test microorganism used was Pseudomonas Aeruginosa. This is the most common disease-causing species, according to the Center for Disease Control and Prevention (CDC).
In this study, Tryptic Soy Agar was used as a positive control. This medium has no antimicrobial efficacy, and is known to support the growth of the test microorganism. The positive control confirmed the test microorganism was viable and pure. The negative control in this study used nutrient salt agar, which does not support bacterial growth. The negative control confirmed that it did not support the growth of the test microorganism. See below for results.

The Boltaron sheet samples did not show any signs of bacterial growth after three weeks.